
Support
Librarians at the University of Westminster can assist with planning, conducting, and reporting systematic reviews and other types of research reviews. To learn more about conducting systematic reviews at University of Westminster, contact your Academic Liaison Librarian.
How to join the University of Westminster Covidence institutional licence:
You can create your personal sign in information with Covidence before or after joining the institutional subscription. To request access to the institutional account in Covidence, you must use your current University of Westminster email address (@my.westminster.ac.uk or @westminster.ac.uk).
Go to - Request your invitation page.
Enter your information using your @my.westminster.ac.uk or @westminster.ac.uk email address and click on “Request Invitation".
Accept the invitation in your email.
Log in if you already have an existing Covidence account with your university email address or sign up for a new account (with your university email).
If you have already joined the University of Westminster Covidence account, then you can log into Covidence and log in with your email and password and proceed to use Covidence.
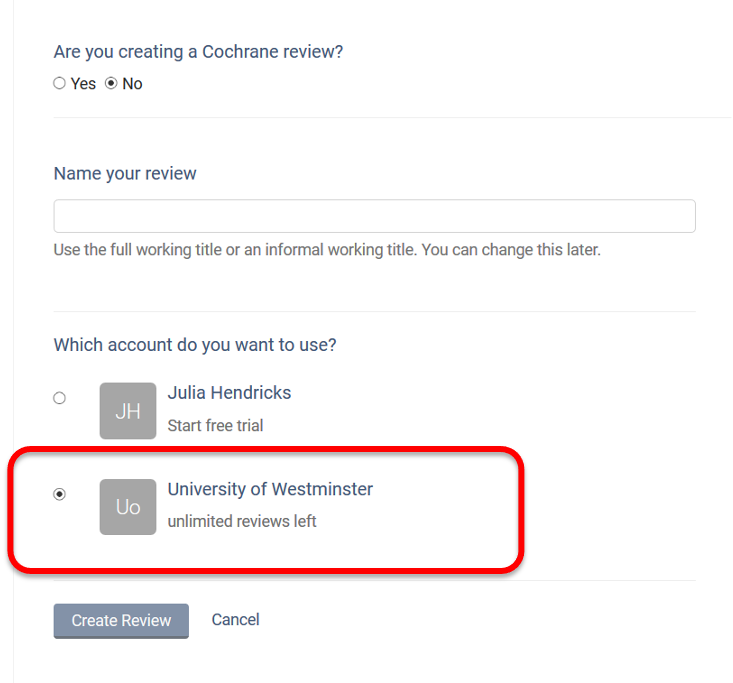
Creating a review using the University of Westminster unlimited licence:
When creating a new review, ensure you choose the University of Westminster institutional licence.
Even if you had a personal account previously, you can now use the University licence.
Note: Reviews created under the institutional license will be visible to the administrators of the University of Westminster Covidence account. Your personal account review(s) will only be seen by you.
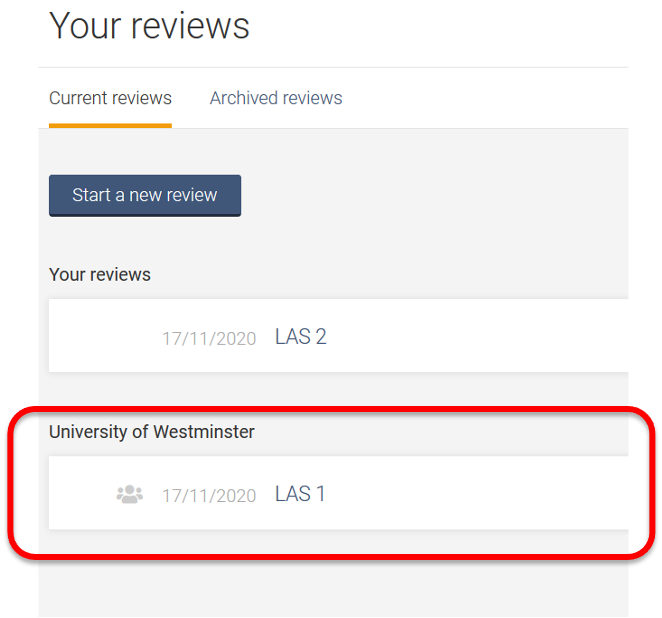
Once you have created a review or accepted an invitation to another University of Westminster account review, the title will appear in a separate section on your account homepage:
Support from Covidence
Covidence offers a range of available resources and support to help you along the way with your systematic review process.
To get familiar with how a Covidence workflow works, you can try our pre-set Demo review. This will allow you to have a hands-on experience and understand the process better. You will find it at the bottom of your dashboard once you have signed in. Give it a go and feel free to explore!

Getting started with Covidence - video tutorials that guide you through the initial area to get you started.
Covidence Knowledge Base - home to all of our comprehensive articles, instructions and advice to FAQ.
Covidence Academy - Learn and understand the essentials of systematic reviews and gain insight with expert-led training and content to guide you through every step of the review process.
Monthly Open Training Webinars - Join our Community Team for free webinars (registration required) or watch our recent recordings without delay.
Online Support Team - contact our team directly for support on your review by emailing support@covidence.org
When logged into your account, you can also easily contact the Support Team and the comprehensive articles from our Knowledge Base within Covidence by clicking the 'Need help?' button in the lower right-hand corner of the screen.

More about Covidence
In Covidence you can:
Import references - Covidence works seamlessly with your favourite reference managers like EndNote, Zotero, RefWorks, Mendeley or any tool that support RIS, CSV or PubMed XML formats. Covidence can automatically remove duplicates for you.
Screen title & abstract - Breeze through screening with keyword highlighting & a lightning-quick interface. Covidence keeps full records of who voted and supports single or dual screeners.
Bulk PDF import - Transfer PDFs stored in your reference manager to Covidence in a few clicks.
Screen full text - Decide quickly on studies in full text. Capture reasons for exclusion and any notes so you can resolve any disagreements quickly, with a click of a button.
Create forms - Be in control and stay focused on your PICO question. Customizable extraction forms mean you only spend time extracting what you need.
Customize risk of bias - Automatically populate your risk of bias tables by highlighting and commenting on the text directly in your PDF.
Conduct data extraction - Extract data efficiently with a side-by-side view of your customized form and PDF. Then, when you are done, easily compare your form with other reviewers.
Export - Covidence exports to all the common formats so you can continue your review in your preferred software.
Collaborate - You can invite other reviewers (including external colleagues) to work with you on the project.

